in Buying Guide, General, Industry
New Guidance on Protein Detection
New guidance was published on the 15th of May, relating to the prevention of CJD and vCJD by Advisory Committee on Dangerous Pathogens’ Transmissible Spongiform Encephalopathy (ACDP TSE) Subgroup. The guidance that has been released In relation to protein detection is potentially huge, in terms of the amount/level of protein that will be allowed on surgical instruments after decontamination, moving forward. If you want to read the full guidance, it can be view here:
Annex C contains the information in relation to protein detection (https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/427855/Annex_C_v3.0.pdf) . A working group was convened to look at the success and outputs of Department of Health funded research project, the aim of which was to extend the evidence available for the decontamination of surgical instruments and the protein detection on these. The working group reviewed new technologies that are available for protein detection, some of which came through Department of Health funded research projects. These new technologies are available so protein detection can be improved in the future on surgical instruments for the well being of anyone who come into contact with surgical instruments in a clinical environment.
These technologies have been previously presented at pertinent congresses, such as the IDSc conference the ProReveal is one of these solutions and it allows an instrument to be scanned for residual protein, facilitating for the first time, quantitative and qualitative results, with a 3D visual showing potential hotspots. It is recommended by the engineering research group that a routine test for washer-disinfectors could be developed to measure the cleaning efficacy at validation and routine testing ‘This method could be based on a process challenge device system that will monitor the optimised wash cycles’. The method, it is proposed, should produce an accurate and consistent set of results that can be assessed by the department and notified bodies where necessary.
Practically, what does this all mean for surgical instruments and CSSD???
Protein levels are used as an indication of the amount of prion protein contamination and it is necessary to check protein levels on instruments after routine reprocessing. Interestingly, this research found that current methods which used swabbing of surgical instruments found that Ninhydrin is insensitive to protein detection and proteins are actually poorly desorbed by swabbing. As an instrument manufacturer, the main thing to come from this study is this staggering recommendation for just how much protein should be allowed/permitted on a surgical instrument
“upper limit of acceptable protein contamination after processing is 5ug BSA equivalent per instrument side”.
A lower level is necessary for neurosurgical instruments! So in terms of everyday surgical instruments, would this be achievable? Work has been done previously by Herve et al which has shown that applying a coating to stainless steel, reduces the ability for protein to stick to instruments, so maybe this is the way forward for new instruments going into production if we are to achieve anywhere near the 5ug level.
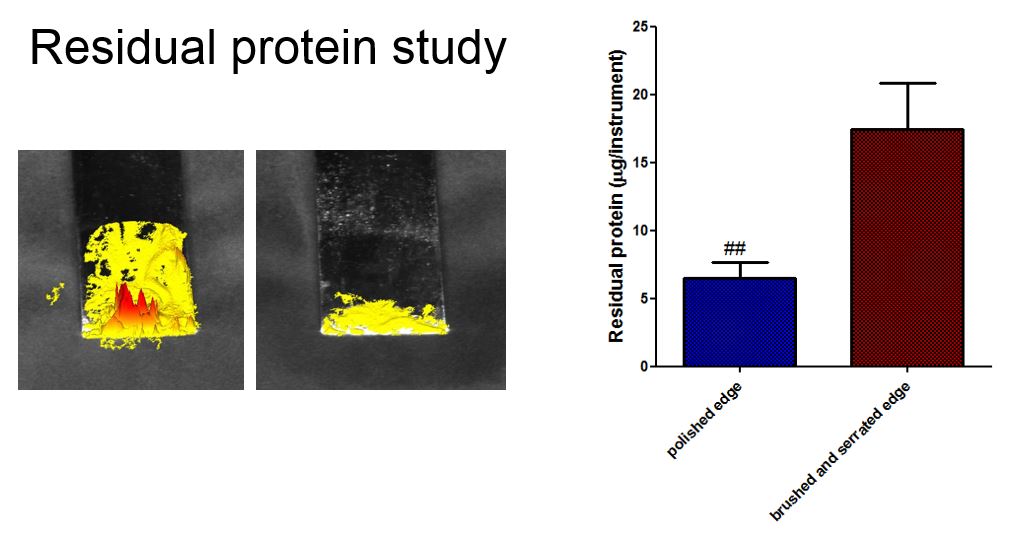
Herve also suggests that surface finish will have an impact on protein retention and it stands to reason that a rougher surface will retain more protein than a smoother surface, stainless steel or not. So this begs the question, what about all the millions and millions of instruments currently in the NHS/Hospital systems. Refurbishment can certainly bring old instruments back to life, surfaces of instruments can be refinished, as the image ‘before and after’ below shows.
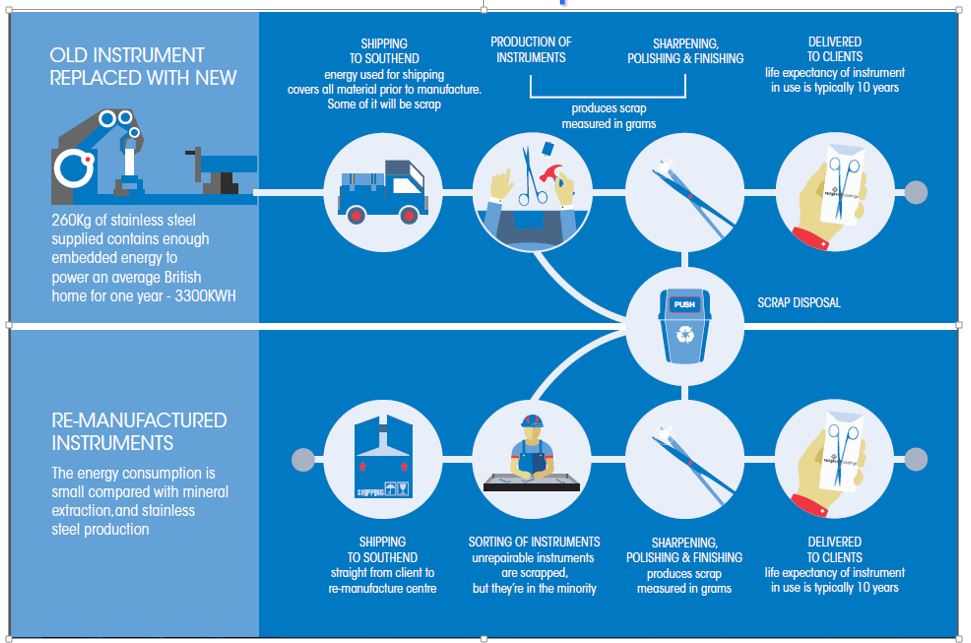
A combination of manufacturing techniques can be used to achieve this and hospitals are opting to refurbish sets, as this not only brings a financial benefit (saves 70% vs purchasing new instruments), but also this also reduces the Carbon footprint of the hospital. The graphic below shows a comparison of 2600 instruments being manufactured from new, vs the same amount of instrument being refurbished; it saves enough energy to power a 2 1/2 houses for a whole year!
We carried out some initial studies on protein retention on rougher surfaces and polished surfaces and initial findings suggest that the polishes surfaces are much improved in terms of protein retention (4X reduction). This is certainly a positive for refurbishing retrospective instrument inventory:
Conclusion
It seems these recommendations may eventually find themselves integrated into the next publication of CFPPs, whether the 5ug is practical or not will remain to be seen, but what is clear is that alongside the recommendations, new technologies such as ProReveal are effective and allow us to look at what can be done to the instruments design wise/coatings to reduce protein adhesion. Refurbishing instruments is certainly a cost effective option and saves a lot of money for the hospital, but the answer could be in improvements to cleaning chemistries/washer disinfectors, as practically these instruments have to be manufactured from stainless steel.










Hello. I’m interested in this study in relation to the surface finish retention properties against the typical surface finishes found in use. Also the critical area is as always, the working surfaces so I’m guessing that these will be either difficult to improve or by doing so we would reduce the usefulness of the instrument by affecting grip, for example. Can I access a copy please?